why is pediatric aml so far behind?
Progress lags in pediatric AML (acute myeloid leukemia) because it is a complex group of diseases, not one, that can vary from patient to patient, even within one subtype. And, because myeloid cells are essential, safely targeting AML has been challenging.
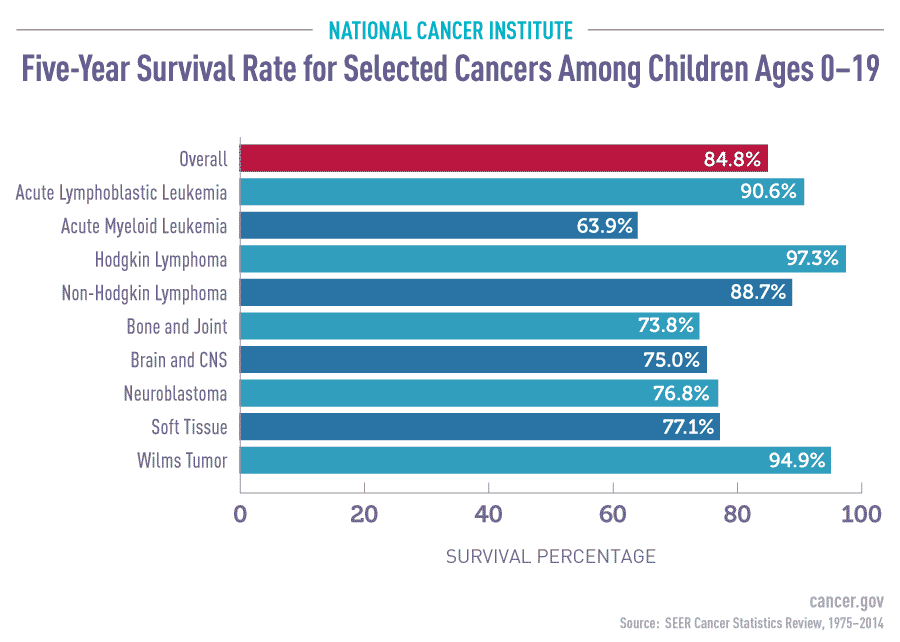
Leukemia is the most common form of childhood cancer. Although it accounts for less than one quarter of all leukemias in children, AML is the deadliest. It remains one of the poorest prognosis pediatric cancers with a notoriously brutal and dangerous treatment regimen that has not fundamentally changed in decades. 5-10% of all young AML patients will die in treatment from treatment-induced complications.
Multiple rounds of high-intensity chemotherapy, full-body radiation, and one or more bone marrow transplants remain the primary therapies for many young AML patients, particularly the 50% who relapse. Survivors face daunting long-term, treatment-related side effects -- diminished organ function/failure, growth issues, infertility, and secondary cancers (to name a few). AML is also the most common form of secondary cancer that can develop in survivors of other pediatric malignancies.
Progress in Acute Lymphoblastic Leukemia (ALL), the most common childhood leukemia, is pushing survival over 90%. CAR T-cell therapies have been a significant breakthrough that may thrust survival near 100% in the coming years as more targets (and CAR-Ts programmed to those targets) are discovered.
But, why so much progress in ALL? Most kids with ALL express a protein called CD-19 on the surface of both their healthy and leukemic B lymphocytes. Other good targets that can be programmed into CAR T-cells exist for ALL. But, today’s CAR T-cells kill both healthy and leukemic cells. This is not a problem in ALL, because you can live a relatively normal life without your B lymphocytes.
Targeting - A challenge and an opportunity
AML is a more complex and diverse disease, so progress lags. Targeting AML without destroying healthy, life-essential myeloid cells remains challenging. And, because AML differs so significantly from patient to patient, one or two “magic bullet” T-cells, or other agents, are unlikely to help everyone. Combining and tailoring therapies, including targeted agents, to the individual patient (based on their genomic profile), is currently the most promising approach to improving outcomes for young AML patients.
Because AML is so complicated, a big study of many patients across all known subtypes is needed to understand the full “disease landscape” of pediatric AML. Target Pediatric AML is a large-scale, data science effort that will examine the genetics of AML across 1000 young patients, looking for unique identifiers, drivers and vulnerabilities that can be specifically and more safely targeted, either by new agents or existing drugs designed for other diseases.